Other references
Researchers User Guide (Part A)
Researchers User Guide (Part B)
In this page...
Submission
1. When all forms and documents for the ethics application are authorised, the CPI or CPI Delegate can select 'Submit to Ethics' button in the Applications tab (do not have to select a form or documents before clicking the 'Submit to Ethics' button).
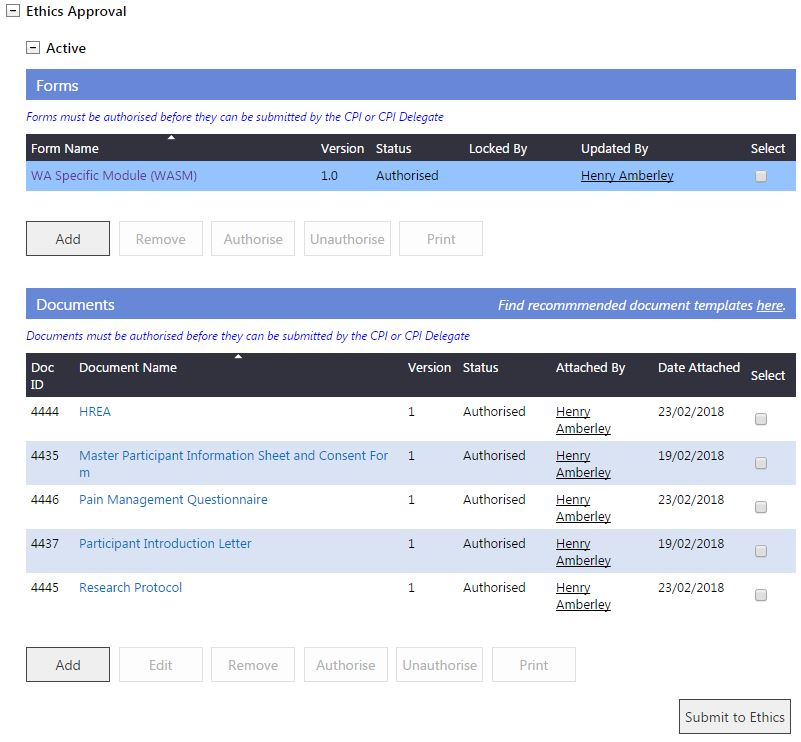
2. The 'Submit Project to Ethics' screen will launch and will display all authorised forms and documents. Select the HREC, and forms and documents for submission. Click 'Submit Project'.
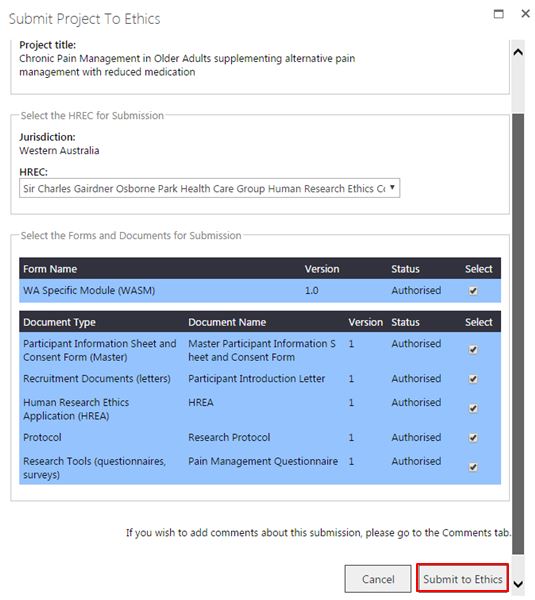
An ethics application requires either a WAHEAF; or a WASM form and a HREA document uploaded to proceed with the submission. The initial ethics application requires the form and documents to be submitted as a package. If the ethics form is not 'Authorised' there will be a message that there are no forms available for submission. If only documents are selected for your initial submission RGS will inform you that an ethics form has to be included in the submission.
3. You will receive a screen message advising the successful submission of your ethics application. The forms and documents will move to 'Submitted' in the Applications tab. Once in the Submitted section the forms and documents cannot be removed or deleted.

Note that you cannot submit another form or document until the validation of any previous submission has been completed.
WA Health Lead HREC (With a WA Health Specialist HREC)
1. If you are submitting to a WA Health Lead HREC and an Additional WA Health HREC (Specialist), you will be able to submit the same forms and documents to both HRECs. Select the Lead HREC and submit the forms and documents as per the steps above.
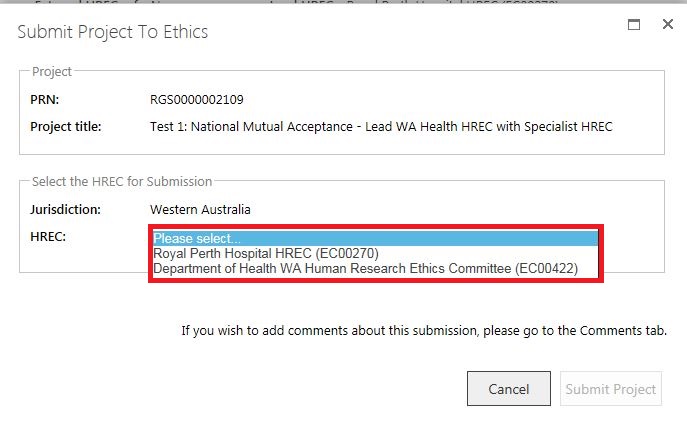
2. The forms and documents will move to the 'Submitted' section of the Applications tab. Click 'Submit to Ethics'.
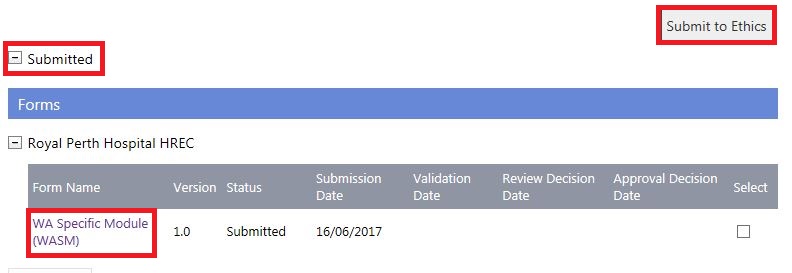
3. The 'Submit Project to Ethics' screen will launch. Select the Specialist HREC, the forms and documents will now be displayed as submitted. Select them and then click 'Submit to Project'.
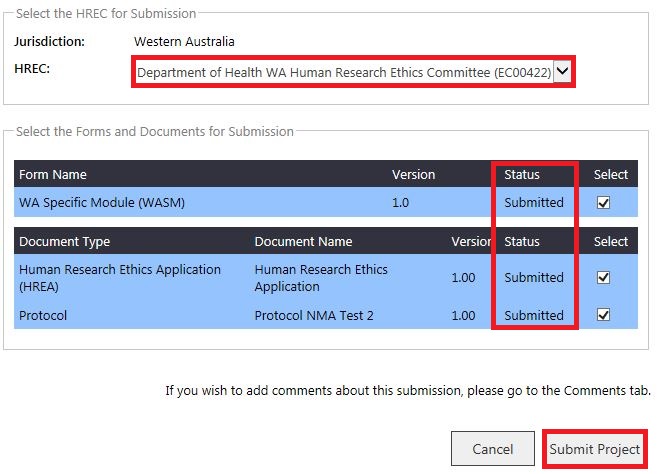
4. The forms and documents will move to 'Submitted' in the Forms & Documents tab and be displayed for both HRECs.
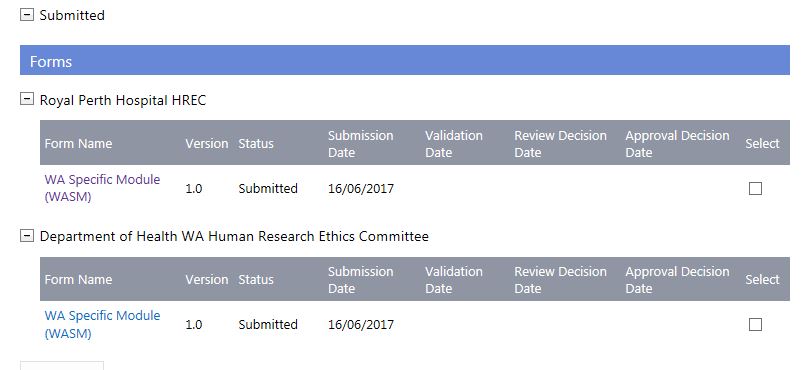
External Lead HREC (With a WA Health Specialist HREC)
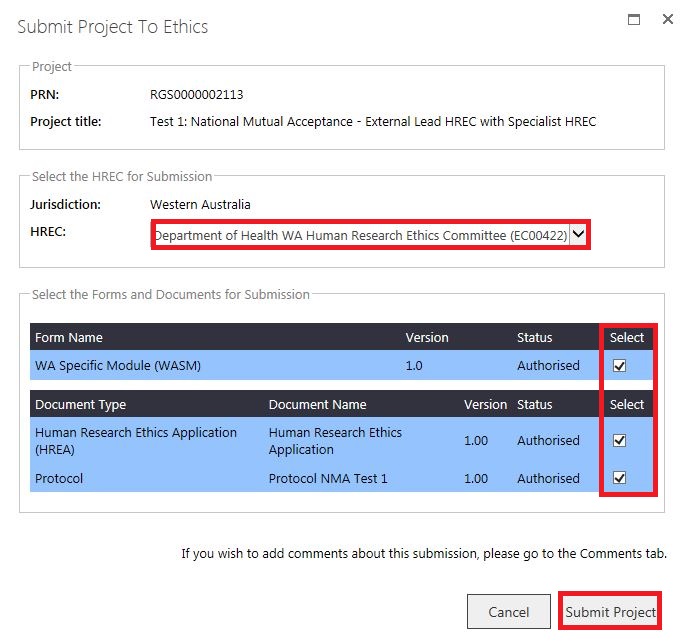
1. For NMA, if an External HREC (outside of WA Heath) is the Lead HREC and you are still submitting the ethics application to an Additional WA Health HREC (Specialist)., you will only be able to select the Specialist HREC as RGS only allows submission of an ethics application to a WA Health HREC.
External Lead HREC Only
1. When the External Lead HREC has approved the ethics application, the approved HREA and supporting documents (including the ethics approval letter) must be uploaded into the Ethics Approval Active section and submit them with the approved WASM to the RG Office.
2. Prior to submission, the WASM and the Documents must be authorised by the CPI or CPI Delegate. The CPI, CPI Delegate, PI or PI Delegate must click "Submit to RG Office' to submit the approved ethics form and documents to the RG Office for their information.
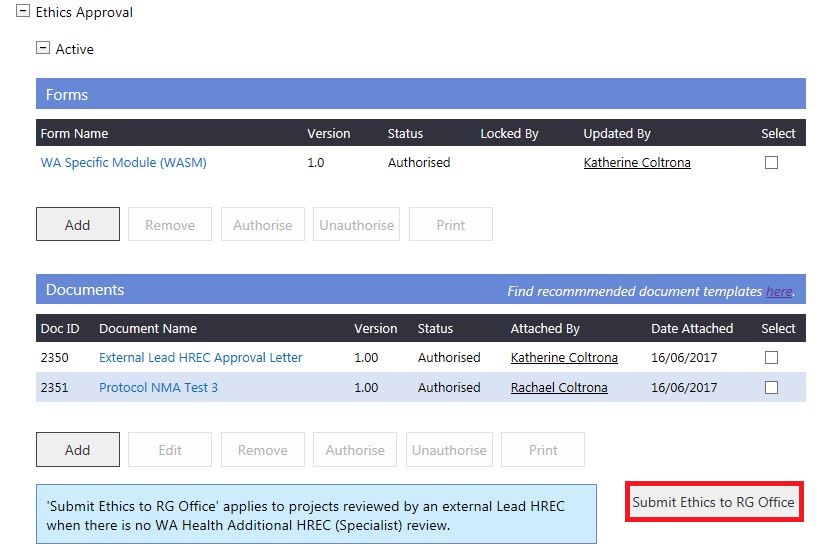
3. The Submit External Ethics to RG Office screen will launch. Select the RG Office, WASM and documents for submission. Click 'Submit Project'.
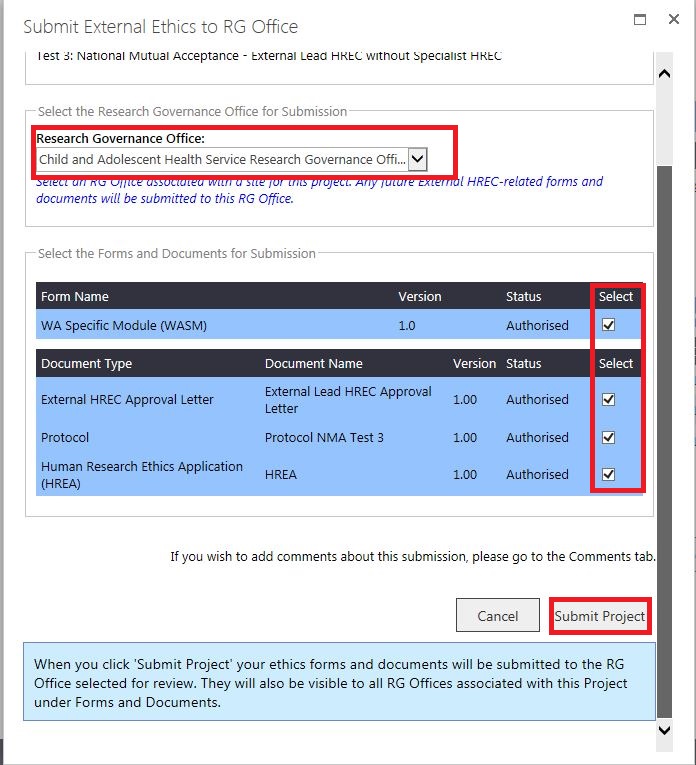
4. The submitted WASM and documents will be displayed under 'Submitted' against the RG Office name and a successful submission notification will display.

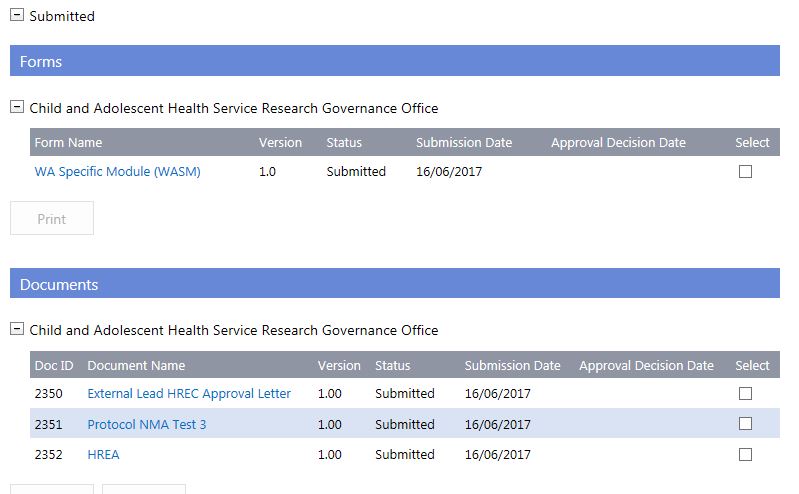
Validation
1. Following the successful submission of the ethics forms and documents, the EEO will validate the submission. Once completed, the CPI and CPI Delegate will receive an email informing them that the submission has been through the validation process and to review the outcome in RGS.
The outcome of the validation process is viewable in the Applications tab in the Submitted section. Click on the link (View) under the 'Validation Date' to open the 'View Validation Assessment' screen.
At any time, you can print or view a Form or Document(s). For forms click on the name link to view or select the form and click 'Print'. For documents, either click on the name link to view or select the document and click 'View'. To print, select the document and click 'Print'.
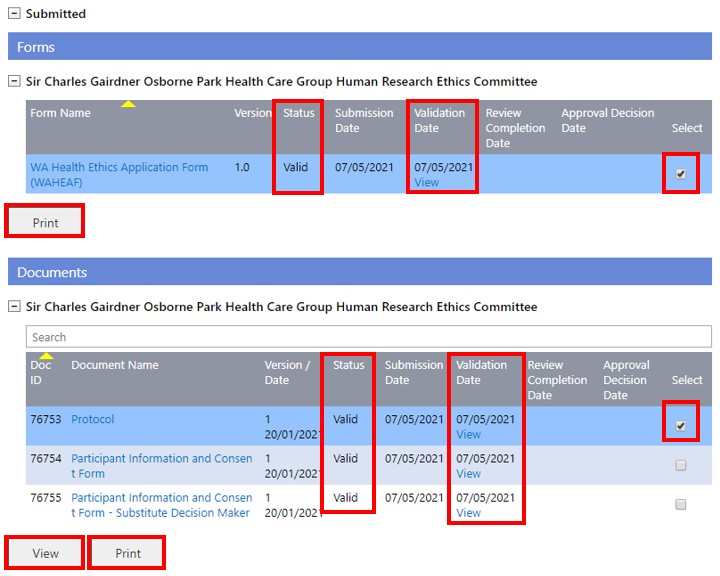
2. The View Validation Assessment screen will launch for you to review the validation status and comments.
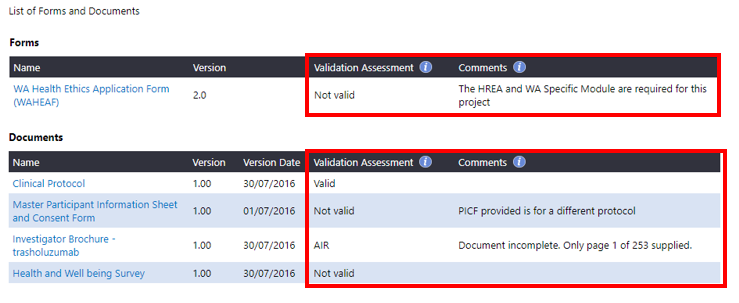
The EEO may decide that
addition information is required (AIR) from the CPI/CPI Delegate if a form or document contains insufficient or incorrect information. The EEO will assign the form or document an 'AIR' status (or Not Valid) and provide information in the 'Comments' box. The CPI/CPI Delegate can read the comment and address this when resubmitting the updated or the correct form/document.
3.
If a form is given the validation status of 'AIR' in the 'Submitted' grid, a new version of the form is created in the 'Active' section of the 'Ethics Approval' (if WAHEAF or WASM). The CPI will need to add the information requested in this new version and submit the form for validation and review.
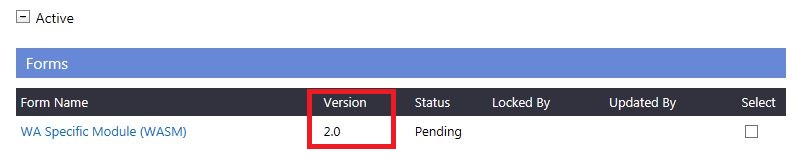 4.
If a document is given the validation status of 'AIR' in the 'Submitted' grid, then the additional information is provided in a new version of document which once uploaded can be submitted for validation and review.
4.
If a document is given the validation status of 'AIR' in the 'Submitted' grid, then the additional information is provided in a new version of document which once uploaded can be submitted for validation and review.
Review Assessment
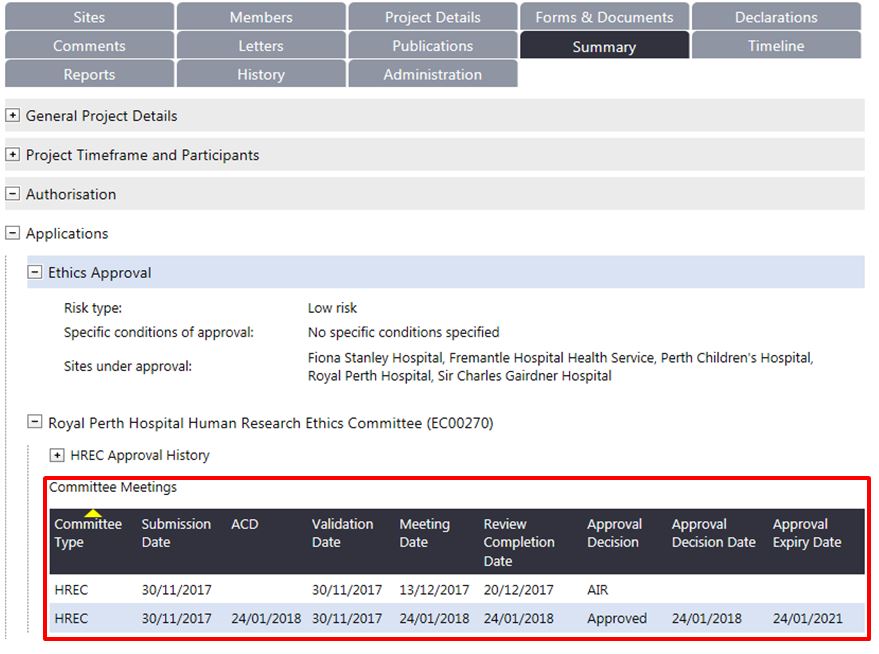
1. If all the forms and documents are valid, they will be added to an ethics committee meeting (Alternative Review, Subcommittee and/or HREC meeting).You can track your project's ethics committee review in the Summary tab:
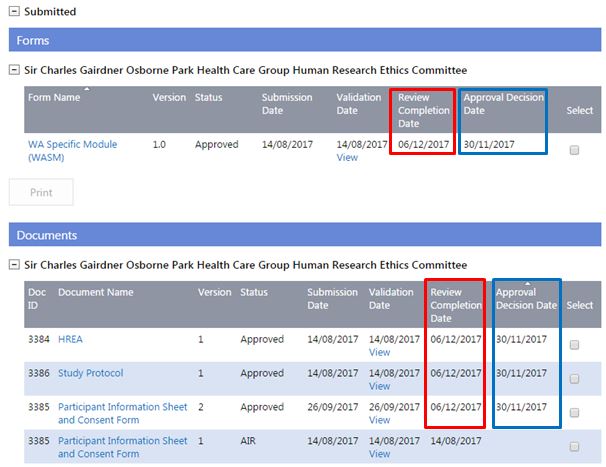
2. You can track the approval of your project's forms & documents in the Forms & Documents tab:
- 'Review Decision Date' is when the committee review is completed and the EEO enters the decision of the review (i.e. generates a letter).
- 'Status' reflects either the validation or committee review decision
- 'Approval Decision Date' is the date the forms and documents were approved at the committee meeting. The Approval Decision Date for the WASM or WAHEAF is the same date as the HREC approval date.
3. Following a review (at an ethics committee meeting), if the Committee requires revisions to a form and has marked it as
'Additional Information Required' (AIR) in the 'Submitted' grid,a new version of the form will automatically be created under the 'Active' grid. You must implement the required changes to this version, which will then be submitted to the Committee for review at the next Committee meeting.
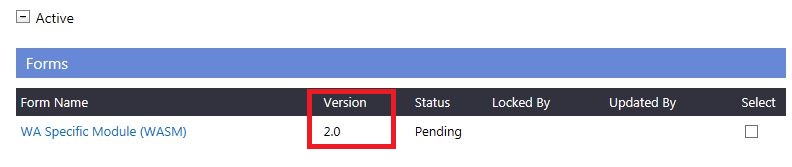
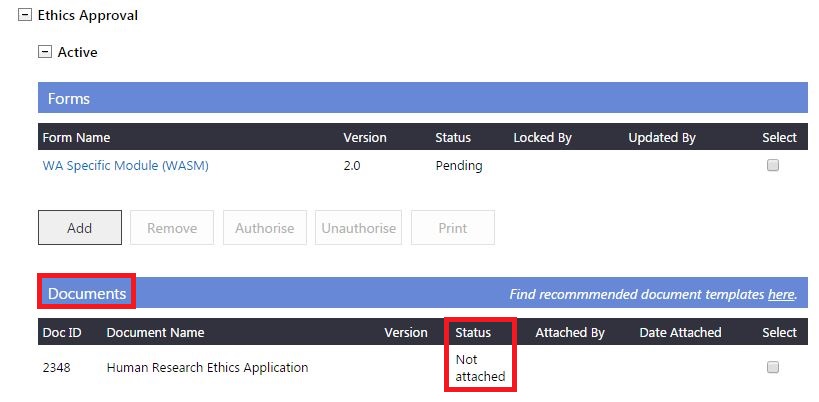
4. Following a review (at an ethics committee meeting) If the Committee requires revisions to a document and has marked it as
'Additional Information Required' (AIR) in the 'Submitted' grid. You must upload the revised document version that addresses the Committee's review comments, which should then be submitted to the Committee for review.
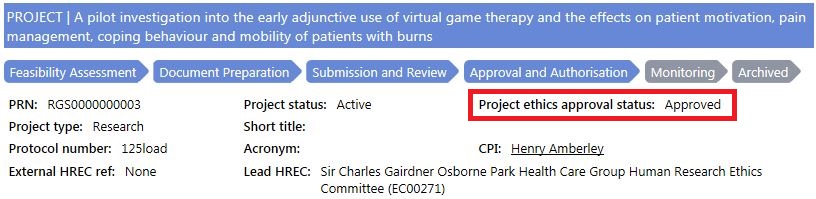
5. The Project ethics approval status of the Lead HREC is displayed in the Project Header:
Other references
Researchers User Guide (Part A)
Researchers User Guide (Part B)