Other references
Researchers User Guide (Part A)
Researchers User Guide (Part B)
All new research projects are allocated their own unique workspace to ensure security of information and an audit log of those accessing the project. The creation of this workspace must be approved by an Administering Research Governance (RG) Office to ensure:
- the validity of the research project i.e. it is a research project conducted within WA Health or accessing WA Health's participants, their tissue or data
- duplicate workspaces are not created for the same project, as project workspaces cannot be merged following creation.
To request the creation of a new project workspace the CPI (i.e. the person submitting the ethics application to the Human Research Ethics Committee (HREC)) must complete and submit a 'Create Project' form. This must be done by the CPI as their name will populate across the project. If you are not the CPI you must not complete the form.
The application is typically processed within 1-2 working days however, actual processing time may vary depending on a range of factors.
National Mutual Acceptance (NMA)
For the NMA scheme a new project workspace must be created by the CPI irrespective of whether the Lead HREC is from WA Health or an external jurisdiction participating in the NMA. This will allow for the CPI or CPI Delegate to either:
- Submit their ethics application to a WA Health Lead HREC and/or Specialist HREC; or
- If submitting to a Lead HREC outside of WA Health:
- Complete the WA Specific Module, print and submit it to the external HREC.
- Upload the approved ethics forms, documents and approval letter to a WA Health RG Office.
Create a new project workspace
1. Click on 'Create Project' to launch the form. It is situated on the left-hand side of the RGS screen.
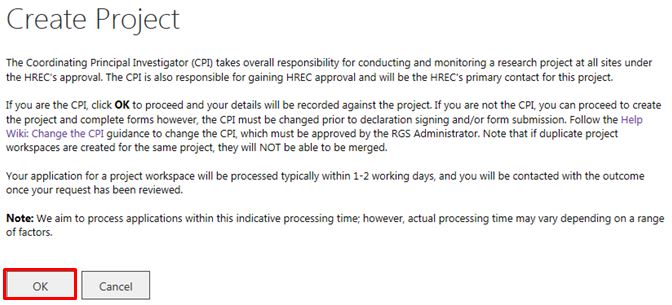
2. A warning message will launch outlining the role of the CPI and the processing of new project requests. If you are the CPI click 'OK' to proceed or 'Cancel' to return to the homepage. The CPI is the person who submits the project to the Lead and/or Specialist HREC for scientific and ethical review and is responsible for the ongoing communication with the reviewing HREC.
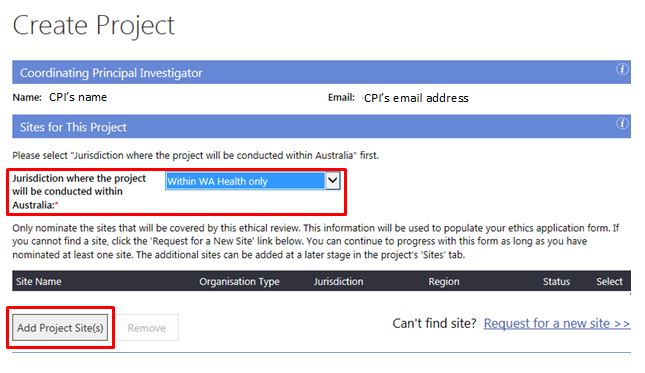
3. The 'Create Project' screen will launch. All mandatory fields have a red asterisk (*) next to them.
Sites for This Project
Select the 'Jurisdiction where the project will be conducted within Australia':
- Within WA Health only
- Intra-jurisdictional (within WA only)
- Inter-jurisdictional (across Australia).
Your answer should reflect all sites where the project is being conducted irrespective of whether the project is reviewed by HRECs outside of WA Health.
The NMA scheme applies to the ethical review of multi-centre research projects conducted across jurisdictions, if the project will be reviewed under NMA, select 'Inter-jurisdictional'.
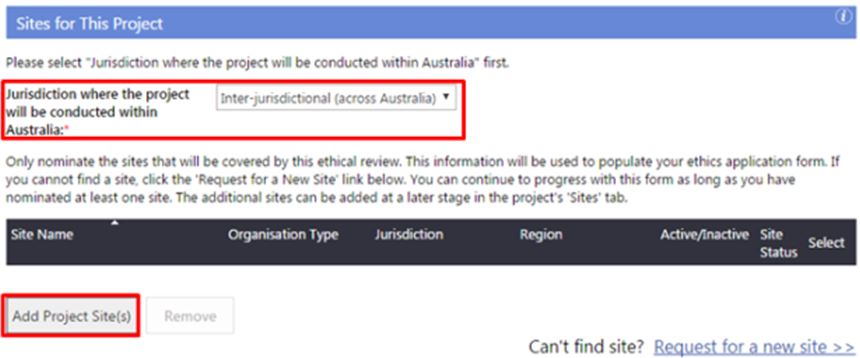
4. Click 'Add Project Site(s)' to launch the Select Site(s) screen.
Only add sites that are covered by Lead or Additional (Specialist) HREC's ethical review as this will be used to populate the Sites tab and WA Health Ethics Application Form. At this time, the Human Research Ethics Application (HREA) cannot be pre-populated.
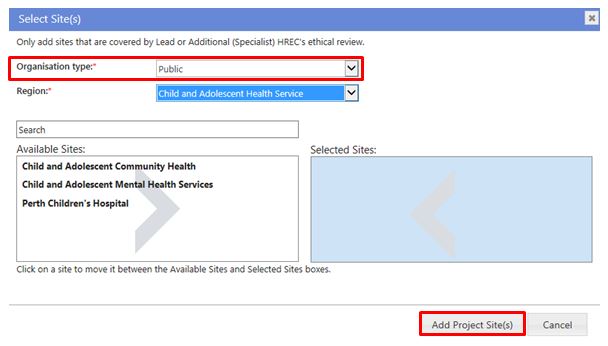
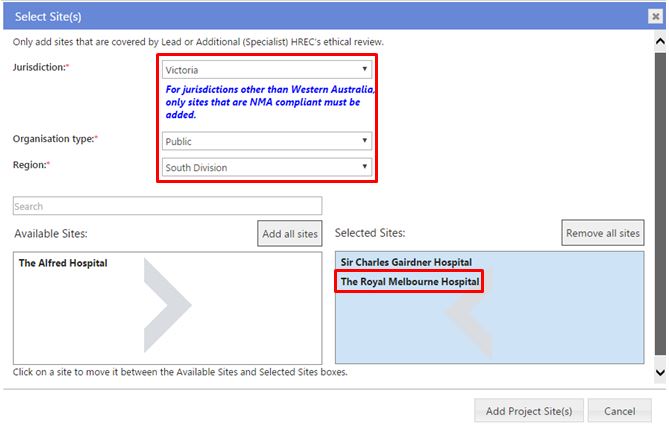
Based on your answer to 'Jurisdiction where the project will be conducted within Australia' you will be asked to select jurisdiction, organisation type and/or region. The organisation types are:
- Public – select the Region and then the site(s)
- Private - select the site(s)
- Research Institutes / Universities – select the site(s).
Select a site by clicking on the site to transfer it from 'Available Sites' to 'Selected Sites', and vice versa. When all the sites have been selected, click 'Add Project Site(s)' to save any changes made, or 'Cancel' to discard them. To remove a site from the Create Project screen, select the site and click 'Remove'.
If you cannot find a site, click 'Request for a new site' located on the left-hand side of the Create Project screen. You can continue to progress with the form as long as you have nominated at least one site. Additional sites can be added later in the project's Sites tab.
User tip: The Department of Health website has further information about the structure and Health Service Providers (HSPs) and the hospitals and health services linked to the HSPs.
For NMA, only add public organisation sites from jurisdictions participating in the NMA.
Project Detail
5. Complete the following:
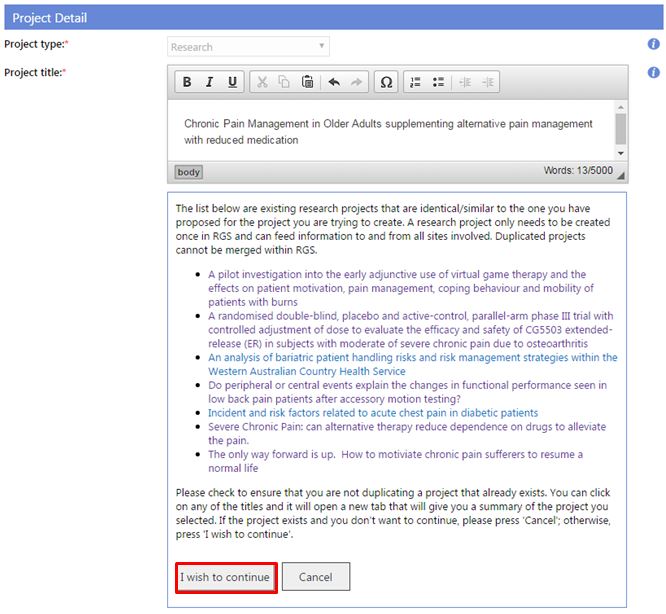
- Project type: this is currently locked to Research projects only. If your project is deemed by the reviewing HREC as 'Quality Improvement' or 'Authorised Prescriber', this can be amended in Project Details – Project Header.
- Project title: this should be the same as the title of the research protocol. Do not bold as this is populated throughout the system.
When you type the Project title, existing projects that are identical/similar will be shown to ensure a duplicate project workspace is not created for an existing project. Once created a project workspace and their associated forms and documents cannot be merged with another project. You can click on any of the titles and it will open a new tab that will give you a summary of the project you selected. If the project exists and you don't want to continue, please press 'Cancel'; otherwise, press 'I wish to continue'.
- Short title: is the lay title that explains the project with a minimum of medical terminology and is usually included on the Participant Information Sheet and Consent Form.
- Acronym: This is the acronym by which the project is referred to, it is not mandatory.
- Keywords: this should be a minimum of three (3) keywords and should be separated by a semi-colon (;). These keywords enable users to accurately search for the project.
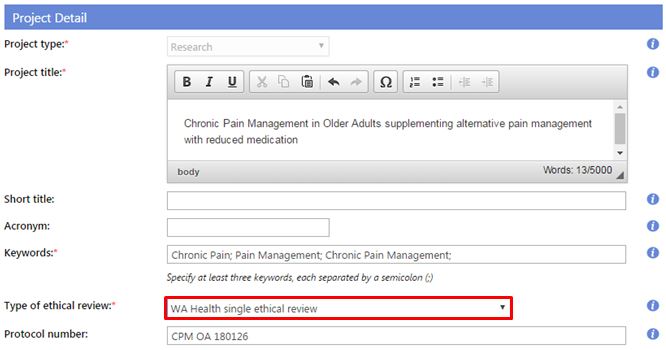
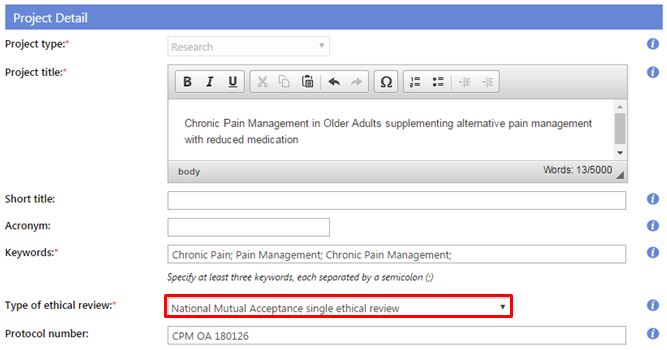
- Type of ethical review: Select either:
1. WA Health single ethical review - The research project is conducted at one or more WA Health sites under the authority of a WA Health HREC (This authority may include an additional ethical review by a specialist WA Health HREC).
2. National Mutual Acceptance (NMA) single ethical review - The research project is conducted at one or more Australian sites under the authority of a NMA certified HREC. (This authority may include an additional ethical review by a specialist NMA HREC).
Protocol number: is the number listed on a Protocol. If there is no protocol number associated with this project, then the version and date of the protocol to be submitted can be used. If required, protocol templates are available for clinical trials and non-clinical trials on the RGS Document Templates page. To meet Good Clinical Practice Guidelines, the protocol should contain, but not be restricted to, the information contained within the applicable protocol template.
Administering Research Governance Office
6. The jurisdiction is currently locked to Western Australia. Select the name of the Administering RG Office to nominate the office to process the form. To ensure minimal delays it is suggested that it goes to the RG Office responsible for a site where you intend to conduct the project.
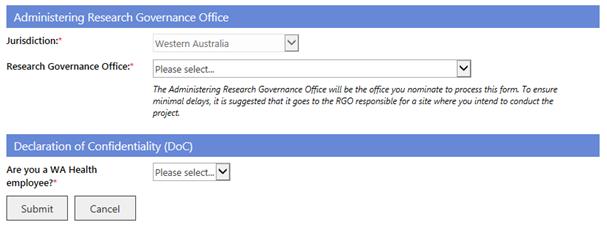
Declaration of Confidentiality (DoC)
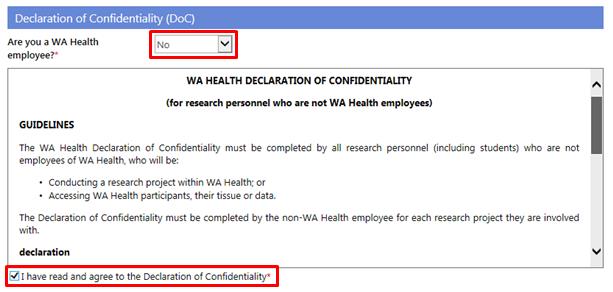
7. All Project Members who are not WA Health employees are required to sign a WA Health Declaration of Confidentiality for each project they are involved with. In addition, if you are a WA Health employee and you are undertaking this project in a non-WA Health employee capacity you must complete the DoC.
Select whether you are a WA Health employee. If the answer is 'No', read the declaration, click 'I have read and agree to the Declaration of Confidentiality' before submitting the form.
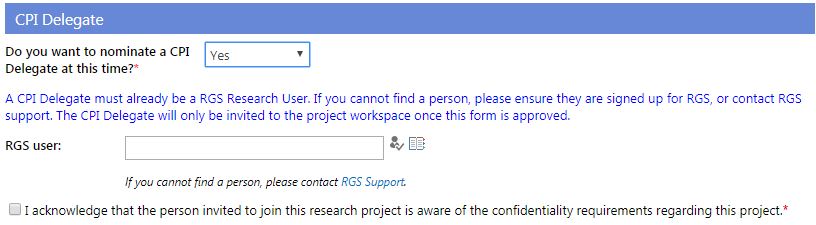
CPI Delegate
8. You can nominate a CPI Delegate when requesting the project workspace. The person you nominate has to be a current RGS user. You can type in the person's name and click the Check Name icon  or to search for the person click the Browse icon
or to search for the person click the Browse icon . When you have selected the person tick the box regarding the confidentiality requirements.
. When you have selected the person tick the box regarding the confidentiality requirements.
9. Click 'Submit' to send the Create Project form to the nominated Administering RG Office for review, or 'Cancel' to discard your changes. If all field validation rules have been met, a confirmation screen will be shown.
10. Applications are typically processed within 1-2 working days by the RG Office and you will receive an email approving or declining your request.

Other references
Researchers User Guide (Part A)
Researchers User Guide (Part B)